New findings on the effects of impurities on catalytic combustion open the way to the production of cleaner energy*
|The catalytic combustion of methane (CH4), the main component of natural gas, is a crucial process for the production of clean energy. Palladium (Pd)-based catalysts are the most active materials for the oxidation of methane at low temperatures. In contact with cerium oxide (CeO2), Pd reaches high activity. The synthesis of Pd nanoparticles surrounded by a thin porous shell of ceria allows full exploitation of this property. However, these catalysts can deactivate under real conditions, especially in the presence of water vapor, sulfur and phosphorus species, as seen in the irreversible deactivation of automotive catalytic converters. The presence of phosphorus compounds (P2O5 or H3PO4) in vehicle exhaust is due to decomposition/volatilization of anti-wear additives that are present in most available motor oils.
Matteo Monai (University of Trieste) and his colleagues shed some light into this matter by performing a fundamental investigation into the mechanism of phosphorous poisoning of catalysts*. The researchers prepared a model catalyst system and exposed it to phosphorous under various conditions. The behavior of the system was studied by a unique combination of techniques, some of which were offered by CERIC: Synchrotron Radiation Photoelectron Spectroscopy (XPS) Scanning Electron Microscopy (SEM), Atomic Force Microscopy (AFM) and X-ray Absorption Near Edge Structure (XANES).
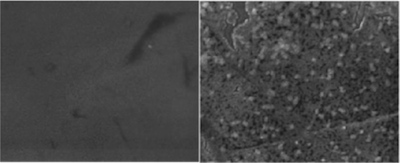
The study led to the conclusion that phosphorous itself only lowers the performance of the catalyst, rather than destroying it completely. Only in combination with water vapor does it lead to the coagulation (clotting) of the CeO2 nanoparticles, which “lock-up” the palladium and completely deactivate the catalyst. An understanding of this mechanism can be used to design more stable catalytic systems, increasing the chance of tackling relevant environmental issues in the future.



